Under our corporate concept “to give first thought to patients and people in the daily living domain, and to increase the benefits that health care provides”, Eisai is committed to improving access to medicines in order to deliver necessary pharmaceuticals to people in need. We strive to create a world where all individuals can equally enjoy a healthy life and realize their fullest lives.
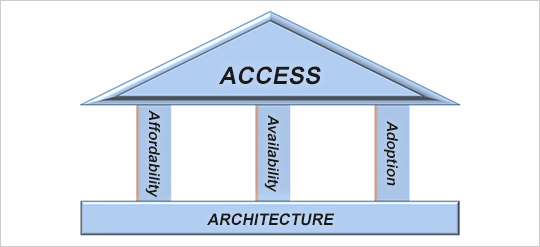
Despite remarkable advances in healthcare and medical treatments in recent years, many diseases still lack effective medicines, and there are regions where essential healthcare services remain out of reach. In fact, due to the lack of appropriate treatment and diagnosis methods, poverty, inadequate healthcare systems, and disparity in access to medical information, more than two billion people unable to receive the care they need or obtain necessary medicines.*1 In light of these circumstances, Eisai believes that it is the mission of modern global R&D-based pharmaceutical companies to create innovative new medicines and contribute to global health and medicine. This means ensuring access to pharmaceuticals—specifically, how we can deliver new drugs that meet medical needs based on the latest life sciences to the people who require them. To improve access to medicines, it is essential to take action from multiple perspectives related to the supply of medicines, including increasing affordability (ease of purchase), availability (opportunity for acquisition), adoption (understanding of the disease and willingness to take medication), and developing the underlying architecture (healthcare infrastructure to support pharmaceutical access).*2 We aim to address the challenges of pharmaceutical access in a comprehensive manner by pursuing the needs of patients and society in each country and region from these four perspectives, while striving for long-term and sustainable solutions.
In particular, we are committed to pursuing and implementing business models for the sustainable provision of pharmaceuticals that are aligned with healthcare systems and economic condition of each country, creating opportunities for access to diagnosis, treatment, and medicines by building disease ecosystems, and improving patient adherence and bridging information gaps to help patients better understand their diseases and treatment plans. Through these efforts, we seek to deliver Eisai’s innovative new medicines to even more people across the world.
The problem of access to medicines, especially in in low- and middle-income countries, cannot be solved by the efforts of a single pharmaceutical company. Rather, it is a global issue that requires cooperation from all sides to solve. Along with governments, international organizations such as the World Health Organization (WHO), other private sector companies, academia and research institutes, nonprofit organizations (NPOs), and non-government organizations (NGOs), Eisai is determined to play a central role in improving access to medicines worldwide through public-private partnerships.
- *1Access to Medicine Foundation “5 Billion People Have Access to Medicine, 2 Billion to Go” Strategic Direction 2017-2019
(Last access, November 30, 2025) - *2Frost LJ, Reich MR, Access: How Do Good Health Technologies Get to Poor People in Poor Countries? Cambridge: Harvard Center for Population and Development Studies, distributed by Harvard University Press, 2008
ACCESS, Affordability, Availability, Adoption, ARCHITECTURE
