- For Print
- September 27, 2012
Eisai Co., Ltd. (Headquarters: Tokyo, President & CEO: Haruo Naito, “Eisai”) today announced that the Eisai Group’s production management structure will undergo a transformation that will see the Group shift from its existing production management structure, which is based on manufacturing sites managed by region, to a new globally integrated unit-based structure organized by product family. By transitioning to the new structure, Eisai aims to clearly define and assign end-to-end accountability for all products, identify the true needs of patients, and further strengthen its production structure so as to provide products that will deliver optimal patient satisfaction.
In fiscal 2010, Eisai’s production headquarters underwent a shift in organizational structure in which it moved away from its “supply-driven” model to a model that is driven by “demand,” and has since been operating as Eisai Demand Chain Systems (EDCS) to carry out production activities aimed at increasing customer satisfaction. The market environment surrounding the current pharmaceuticals industry is undergoing rapid change. Amidst such fast-paced environmental changes, which include the increased importance of emerging markets, challenges regarding access to medicines, the diversification of customer needs due to globalization, and accommodating the production of new technologies such as biologics, EDCS is now shifting to a new structure to identify and respond to the real needs of our customers.
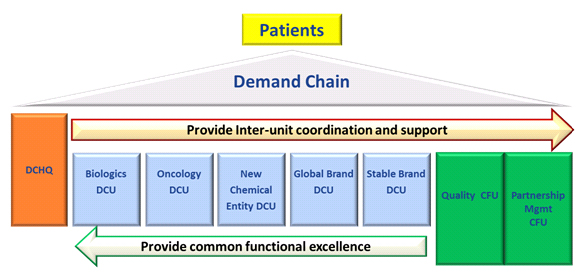
In consideration of Eisai’s product portfolio and technology strategies, the new structure will comprise five product family-based Demand Chain Units (DCUs) and two Core Function Units (CFUs), with each unit autonomously managing their respective activities. Within each DCU, a “Product Champion” will be also assigned to every product. Product Champions will be accountable for all production activities related to their assigned product or products, from procurement of raw materials to production, packaging and distribution, taking ownership of these activities to ensure that each DCU delivers products that provide customer satisfaction. Meanwhile, the CFUs will carry out functions common across all units, including quality assurance and managing contract manufacturing of products, providing expertise in these areas to support the activities of the DCUs. Additionally, a Demand Chain Headquarters (DCHQ) will be established to carry out strategic planning, organizational and talent management and risk management functions to optimize synergies across the entire EDCS organization while maintaining the autonomy of each individual unit.
Under this new structure, Eisai aims to respond to diversifying customer needs in an era of rapid globalization to provide high-quality medicines at affordable prices and deliver products that create customer joy. In doing so, Eisai will continue to make contributions to increase the benefits provided to patients and their families worldwide.
[ Please refer to the following notes for information on each unit and its respective role. ]
Media Inquiries:
Public Relations Department,
Eisai Co., Ltd.
+81-(0)3-3817-5120
< Notes to editors >
1.Overview of Each Unit and Its Respective Role
Biologics DCU:
The Biologics DCU will be responsible for building a facility capable of carrying out scale-ups during the development stage as well as initial commercial production, in preparation for the establishment of in-house production technology and functions for biologics, in particular antibody drugs. (Franchise: Kashima, Japan)
Oncology DCU:
The Oncology DCU will be in charge of the production of all anticancer agents, including Halaven® and lenvatinib. Eisai’s North Carolina-based U.S. production plant will be positioned as the production hub for the formulation of anticancer agents, with the unit working to enhance in-house anticancer agent production capabilities for injectable drug products and solid dose formulations. (Franchise: North Carolina, U.S.)
New Chemical Entity DCU:
The New Chemical Entity DCU will be in charge of the establishment of production technologies and logistics frameworks for new products (with the exception of anticancer agents and biologics) and carry out initial commercial production for such products. Once the production technology for commercial production of a given product has been established, the unit will transfer the technology to either the Global Brand DCU or the Stable Brand DCU. (Franchise: Hatfield, U.K.)
Global Brand DCU:
The Global Brand DCU will be responsible for the manufacture of Aricept®, Pariet®, Zonegran® and other global products. The unit will also be tasked with developing and executing procurement strategies that will enable the Eisai Group to offer its products at affordable prices. (Franchise: Vizag, India)
Stable Brand DCU:
The Stable Brand DCU will be in charge of long-listed drugs and consumer healthcare products marketed primarily in Japan and other parts of Asia. The unit will aim to generate customer joy by spurring further demand innovation within the product group for which it is responsible. (Franchise: Misato, Japan)
Quality CFU:
The Quality CFU will be accountable for quality assurance of all Eisai products sold throughout the world. The structure will comprise four regional functions—the Americas, EMEA (Europe, the Middle East and Africa), Asia (East Asia and the Indo-Pacific region, excluding Japan) and Japan—and one global function. (Franchise: Global Headquarters, Tokyo, Japan)
Partnership Management CFU:
The Partnership Management CFU will be in charge of managing the purchasing and inventory of purchased prescription drug stock, such as in-licensed products, as well as managing the contract manufacturing of products under the jurisdiction of each DCU. (Franchise: North Carolina, U.S.)