- For Print
- May 17, 2011
Eisai Co., Ltd. (Headquarters: Tokyo, President & CEO: Haruo Naito, “Eisai”) announced today that it has received approval from Swissmedic, the Swiss Agency for Therapeutic Products, for Halaven® (eribulin mesylate), in the monotherapy treatment of patients with locally advanced and metastatic breast carcinoma with progression after prior therapy with an anthracycline, a taxane and capecitabine.
The company submitted New Drug Application based on the results from a Phase II study (Study 211) of Halaven® in order to deliver the medicine to patients as quickly as possible. Swissmedic has been reviewing the application based on Study 211 along with the global Phase III EMBRACE study (Eisai Metastatic Breast Cancer Study Assessing Treatment of Physician’s Choice (TPC) vs Eribulin E7389). The application to Singapore has been approved in February 2011.
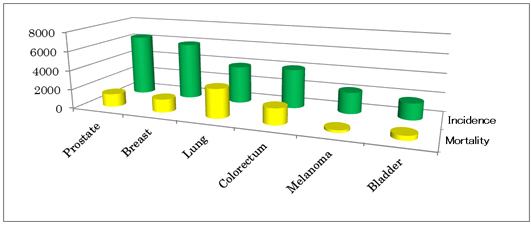
The Swismedic approval means that patients in Switzerland will soon be able to benefit from this innovative treatment. Breast cancer is the second most commonly diagnosed cancer worldwide and there are about 1.3 million new cases of the disease annually. More than 5,000 Swiss women have the disease and approximately 1,400 are likely to die annually1), 2).
Having already received approval in the United States in November 2010, in Singapore in February 2011, in March 17, 2011 in European Union and in April in Japan, the approval of Halaven® by Swissmedic marks the fifth country or region for which the drug has been approved. Halaven® is also currently under regulatory review in Canada.
True to its human health care (hhc) mission, Eisai remains committed to patients and their families and continue its stated goal of delivering Halaven® to patients worldwide as quickly as possible.
[ Please refer to the following notes on the 211 Study, EMBRACE study and Breast Cancer in Switzerland ]
Media Inquiries:
Public Relations Department,
Eisai Co., Ltd.
+81-(0)3-3817-5120
< Notes to editors >
1)211 Study
Study 211 is a Phase II, open-label, single-arm study evaluating the efficacy and safety of Halaven® in patients with locally advanced or metastatic breast cancer who had received an anthracycline, a taxane and capecitabine as prior therapy, and who were refractory to their last chemotherapy regimen, as documented by progression on or within six months of that therapy. Of 299 patients enrolled in the study, 291 were treated with Halaven®. Two-hundred sixty-nine patients met the key inclusion criteria. Investigator-assessed ORR was 14.1% (1 CR). Nearly half (46.5%) the patients had stable disease after treatment with Halaven®. The clinical benefit rate was 17.1%. The median duration of response was 4.2 months. Median progression-free survival was 2.6 months, and the median overall survival rate was 10.3 months. The six-month PFS and OS rates were 16.0% and 72.3%, respectively.
The safety analysis included all 291 patients who received treatment with Halaven®. The most frequently reported Grade 3 or Grade 4 adverse events were neutropenia ,54%, febrile neutropenia, 5.5%, leukopenia,14%, and weakness/fatigue (10%; no Grade 4 events). Grade 3 peripheral neuropathy was reported in 5.5% of patients. No Grade 4 peripheral neuropathy events were reported.
2)About the EMBRACE Study
EMBRACE was an open-label, randomized, global, multi-center, parallel two-arm study designed to compare overall survival in patients treated with Halaven® versus a Treatment of Physician’s Choice (TPC). The study included 762 patients with locally recurrent or metastatic breast cancer who previously had been treated with at least two and a maximum of five prior chemotherapies, including an anthracycline and a taxane. Patients were randomized at a ratio of two-to-one to receive either Halaven® or TPC. TPC was defined as any single-agent chemotherapy, hormonal treatment or biologic therapy approved for the treatment of cancer; or palliative treatment or radiotherapy administered according to local practice. The vast majority (97 percent) of patients in the TPC arm received chemotherapy.
The most common adverse reactions (incidence greater than or equal to 25 percent) among patients treated with Halaven® were asthenia (fatigue), neutropenia, anemia, alopecia (hair loss), peripheral neuropathy (numbness and tingling in arms, legs and other parts of the body), nausea and constipation. The most common serious side effects reported in patients receiving Halaven® were neutropenia with or without fever (4 percent and 2 percent, respectively). The most common adverse reaction resulting in discontinuation of treatment with Halaven® was peripheral neuropathy (5 percent).
3)Breast Cancer in Switzerland3)
In Switzerland, 5,918 were diagnosed as the breast cancer and 1,367 have died in 2008.
- 1)
Estimates of worldwide burden of cancer in 2008: GLOBOCAN 2008, Int. J. Cancer: 127, 2893–2917 (2010)
( http://onlinelibrary.wiley.com/doi/10.1002/ijc.25516/full )
)
- 2)
- 3)
Estimates of the cancer incidence and mortality in Europe in 2006, Annal of Oncology 18: 581 (2007)
