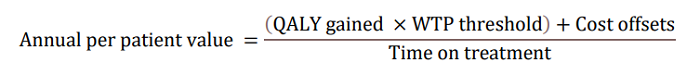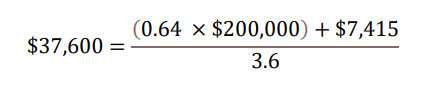- For Print
- January 7, 2023
Based on hhc (human health care) concept, our corporate philosophy, Eisai is committed to improve patients health outcomes and quality of life, simplify care delivery, increase health system efficiency and spur future investments in Alzheimer’s disease (AD). We consider a holistic approach in assessing value and making decisions that may affect patient access, so that our LEQEMBI pricing approach can maximize value for all stakeholders (patients, families, caregivers, healthcare providers, payers, employees and shareholders). This approach includes clinical outcome assessments of our medicines and the benefits we deliver to patients, their families and caregivers as the “clinical value,” as well as the projected “social value” that help improve patients’ and caregivers’ quality of life and productivity. Moreover, we assess the simulated impact of our medicines on reducing demand for health services and global burden of disease as potential “economic value” while enhancing further innovations in AD.
Social Impact of AD in the U.S.
According to the Alzheimer’s Association’s “2022 Alzheimer’s Disease Facts and Figures”1, an estimated 6.5 million Americans aged 65 and older are living with dementia due to AD (i.e., mild, moderate and severe dementia stages of AD). AD was officially listed as the sixth-leading cause of death in the U.S. in 2019 and the seventh-leading cause of death in 2020 and 2021. It is a chronic, progressive, disabling and fatal disease. According to another report from the Alzheimer’s Association, “Changing the Trajectory of Alzheimer’s Disease: How a Treatment by 2025 Saves Lives and Dollars”2, the total costs of care in the U.S. from all payers (i.e., Medicare, Medicaid, out-of-pocket and other payers), would increase from $267 Billion in 2020 to $451 Billion in 2030 if no treatment exists to delay the disease.
A recent 2022 study, “Projecting the Long-term Societal Values of Disease-Modifying Treatment for Alzheimer’s Disease in the United States”3, quoted as many as 10 to 14 million Americans living with MCI (Mild Cognitive Impairment) of any etiology, the very mild symptomatic stage before onset of dementia stages, in which 55% have AD as the underlying pathology. In this study, the lifetime value of a disease-modifying treatment in Early AD (MCI due to AD and mild dementia stage of AD) from a U.S. societal perspective assuming a treatment effect of 30% relative decline in progression rates from MCI to mild dementia and from mild to moderate dementia was estimated at $134,418 per person.
Value of LEQEMBI Adoption to U.S. Society
LEQEMBI is indicated for the treatment of AD, and treatment with LEQEMBI should be initiated in patients with MCI or mild dementia stage of AD after confirmation of amyloid beta pathology (Early AD). In the U.S., we estimate that the diagnosed eligible Early AD population will reach approximately 100,000 individuals by year 3 representing a measured initial attainment in the real world and will increase gradually over the mid-to-long term given the time required to advance new screening and diagnostic technologies such as blood-based biomarkers to confirm amyloid beta pathology.
Published findings in a peer-reviewed journal4 from the confirmatory Phase 3 Clarity AD study in patients with Early AD demonstrate that LEQEMBI treatment resulted in less decline on measures of cognition and function than placebo at 18 months (27% slowing over 18 months measured by CDR-SB*) and was associated with adverse events that were within expectation. Clarity AD results were consistent with that from the Phase 2 trial (Study 201)5 that was the basis for the FDA’s accelerated approval and will be submitted to the FDA very shortly for review for traditional approval.
LEQEMBI’s clinical data show that it could help patients maintain cognition, preserve activities of daily living and maintain functional ability for longer, and therefore, LEQEMBI’s clinical efficacy could potentially translate into impactful outcomes for these patients and their families. A simulation study using an established, validated disease model called Alzheimer’s disease Archimedes condition event (AD ACE),6,7,8 which is an individual patient-level model with a focus on predicting the trajectory of cognitive decline and simulating the effects of early interventions in AD, helped assess the potential lifetime value and economic impact of LEQEMBI in patients with Early AD based on clinical trial populations and findings. The results9,10 of this simulation study based on Study 201 of LEQEMBI were published in a peer-reviewed journal, and the model has been recently updated with data from the Clarity AD trial showing consistent outputs.
Per Patient Societal Value of LEQEMBI at $37,600 per Year in the U.S. by AD ACE Model
In the updated AD ACE simulation study using Clarity AD data, LEQEMBI treatment was projected to delay disease progression, resulting in an increase in patient’s expected time in Early AD while reducing the time in more advanced severe states. Slowing of clinical decline in patients treated with LEQEMBI is estimated to delay disease progression by nearly 3 years on average (delay progression mainly from MCI to mild AD and from mild to moderate AD) compared to standard-of-care (SOC). LEQEMBI’s impact on the disease trajectory is then modeled into an annual per-patient value to the U.S. society based on four components according to the following equation: a) quality-adjusted life-years (QALY) gains compared to SOC; b) willingness-to-pay (WTP) threshold; c) cost offsets compared to SOC; and d) time on treatment, all in present value term. Note that the costs, benefits and time on treatment need to be adjusted for the values between present time and future times using discounting in a health economic evaluation.
With regard to a) QALY gains, since health is a function of length of life and quality of life, this measure combines both attributes into one single index, with one QALY gain representing one additional year of a person’s life at perfect health. LEQEMBI treatment was predicted to offer an additional 0.64 QALYs compared to SOC for an Early AD patient over lifetime by improving outcomes for both the patient and caregiver. With regard to b) WTP threshold per QALY gained, conventionally, the WTP threshold is based on 1 to 3 times country’s per capita gross domestic product (GDP). In the U.S., as a result, a WTP threshold of $50,000 to $150,000 is referenced as the cost-effectiveness threshold11, but a higher WTP threshold is often considered to account for the gravity of conditions with greater burden, and interventions that exhibit wider societal benefits, such as AD with substantial impacts on caregivers. A modified societal perspective at $200,000 WTP threshold per QALY gained is used in the U.S. when societal cost of the disease is large such as AD. With regard to c) cost offsets, avoidance of both direct medical/non-medical costs for AD management, as well as indirect costs of caregivers including family members totaling $7,415 over the lifetime for each Early AD patient, was predicted with LEQEMBI treatment compared to SOC. Direct costs contain cost of medications, medical visits, hospitalizations, living accommodations and community services for the patients. Indirect costs of caregivers, including family members, consider the monetary value for hours spent on caregiving activities. With regard to: d) time on treatment, LEQEMBI was modeled to be stopped upon transition to moderate AD dementia or worse. The average treatment duration in this Early AD population was estimated to be approximately 3.6 years with consideration of discounting to present value.
Taking all four components together in present value term, for a) 0.64 QALYs gained b) at WTP threshold of $200,000 per QALY gained plus c) cost offsets of $7,415 over d) 3.6 years on treatment, the yearly per-patient value of LEQEMBI from a societal perspective was quantified at approximately $37,600.
This yearly estimate of $37,600 equals to approximately $135,000 (0.64 × $200,000 + $7,415) in lifetime value per patient to the U.S. society. Over 10 years cumulatively, the gradual adoption of LEQEMBI treatment among Early AD patients could potentially generate positive social impact of several tens of billion dollars to the U.S. society, in the form of “clinical value” that help patients delay their disease progression, projected “social value” that help improve patients’ and caregivers’ quality of life and productivity, and simulated “economic value” that help reduce demand for health services.
LEQEMBI U.S. Launch Pricing at $26,500 per Year
While we estimate the per-patient-per-year value of LEQEMBI treatment to the U.S. society to be $37,600, Eisai decided to price LEQEMBI below quantified societal value at the wholesale acquisition cost (WAC) of $26,500 per year (estimated annual price based on 10mg/kg IV biweekly for average U.S. patient weight of 75kg based on Study 201 and Clarity AD) aiming to promote broader patient access, reduce overall financial burden, and support health system sustainability. As such, the WAC for the 200mg vial is $254.81 and the WAC for the 500mg vial is $637.02. Actual annualized pricing may vary by patient. In addition, Eisai continues to pursue a less frequent maintenance dosing regimen for LEQEMBI, such as monthly instead of current biweekly regimen, upon significant amyloid beta clearance to prevent re-accumulation of amyloid beta biomarkers while maintaining clinical efficacy. This could further lower the yearly cost of LEQEMBI during the maintenance dosing phase, for example, from $26,500 to potentially about half of this figure given less amount of drugs.
Patient Affordability
Eisai believes patient affordability must be a key consideration to promote patient access and intended use and benefits of LEQEMBI. Among the eligible Early AD patient population in the U.S., once the patient's insurer covers LEQEMBI, we estimate that approximately 91% of individuals will be covered by Medicare with Medigap (supplemental insurance), Medicare Advantage (Medicare-approved plans from private companies with potential supplemental coverage), Medicaid, and Commercial (private insurance12). For these patients, their out-of-pocket costs for LEQEMBI treatment could range from $0 to a few dollars per day. Remaining 9% of the individuals will fall into the category of Medicare without supplemental insurance, and hence will be responsible for 20% of the LEQEMBI cost as co-insurance under Medicare Part B. For these patients, their estimated out-of-pocket costs for LEQEMBI at the price of $26,500 per year will translate into about $14.50 per day13. Across the entire eligible Early AD patient population, we estimate the weighted average out-of-pocket costs for LEQEMBI to be about $2 per day12.
Commitment to Patient Access
Eisai is committed to ensuring that certain financially disadvantaged patients have access to LEQEMBI. Firstly, Eisai is establishing a Patient Assistance Program, which will provide LEQEMBI at no cost, for eligible uninsured and underinsured patients, including Medicare beneficiaries, who meet financial need and other program criteria. Secondly, Eisai will offer patient support for improving access through LEQEMBI Patient Navigators, who will provide information about accessing LEQEMBI, help patients and their families understand their insurance coverage and options, and identify financial support programs for eligible patients.
Health System Sustainability
We believe our pricing approach for LEQEMBI would also help improve health system sustainability, which is projected based on appropriate use of LEQEMBI in eligible patients with Early AD to improve patient’s health outcomes and quality of life and reduce demand for health services and global burden of disease through changing disease trajectory. Furthermore, we believe our pricing approach for LEQEMBI, coupled with the size of the targeted patient population, will be sustainable under historical growth and spending assumptions for Medicare Part B.
Giving Back More Than Half of LEQEMBI Value to U.S. Society
The price of LEQEMBI at a yearly cost of $26,500 is $11,100 below the projected societal value of $37,600, and a less frequent maintenance dosing regimen will further lower the yearly cost well below the projected societal value. Taking these savings as well as discounts and rebates within the U.S. healthcare system into consideration, over 10 years cumulatively, the gradual adoption of LEQEMBI treatment at this pricing approach could give back about 60% of the potential positive social impact of several tens of billion dollars to the U.S. society14. These resources could help realize new innovations that enhance the health and quality of life of individuals at risk of developing or living with AD as well as their families and caregivers. On the other hand, about 40% of the potential positive social impact of several tens of billion dollars will be accrued by employees and shareholders in the form of product sales, from which we are committed to re-invest in future research and development to create new AD therapies and new innovations such as establishing ecosystems toward inclusive AD communities14. We deeply believe that our pricing approach to maximize value for all stakeholders will help Eisai achieve social good in the form of relieving anxiety over health and reducing health disparity according to our corporate philosophy.
*Clinical Dementia Rating Sum of Boxes
(End of Document)
Notes to Editors
INDICATION, DOSAGE AND ADMINISTRATION, AND IMPORTANT SAFETY INFORMATION IN THE U.S.
INDICATION
LEQEMBI is indicated for the treatment of Alzheimer’s disease. Treatment with LEQEMBI should be initiated in patients with mild cognitive impairment or mild dementia stage of disease, the population in which treatment was initiated in clinical trials. There are no safety or effectiveness data on initiating treatment at earlier or later stages of the disease than were studied. This indication is approved under accelerated approval based on reduction in amyloid beta plaques observed in patients treated with LEQEMBI. Continued approval for this indication may be contingent upon verification of clinical benefit in a confirmatory trial.
IMPORTANT SAFETY INFORMATION
WARNINGS AND PRECAUTIONS
Amyloid Related Imaging Abnormalities
- LEQEMBI can cause amyloid related imaging abnormalities-edema (ARIA-E) and -hemosiderin deposition (ARIA-H). ARIA-E can be observed on MRI as brain edema or sulcal effusions, and ARIA-H as microhemorrhage and superficial siderosis. ARIA is usually asymptomatic, although serious and life-threatening events, including seizure and status epilepticus, rarely can occur. Reported symptoms associated with ARIA may include headache, confusion, visual changes, dizziness, nausea, and gait difficulty. Focal neurologic deficits may also occur. Symptoms associated with ARIA usually resolve over time.
ARIA Monitoring and Dose Management Guidelines
- Obtain recent (within one year) brain magnetic resonance imaging (MRI) prior to initiating treatment with LEQEMBI. Obtain an MRI prior to the 5th, 7th, and 14th infusions.
- Recommendations for dosing in patients with ARIA-E and ARIA-H depend on clinical symptoms and radiographic severity. Depending on ARIA severity, use clinical judgment in considering whether to continue dosing, temporarily discontinue treatment, or permanently discontinue LEQEMBI.
- Enhanced clinical vigilance for ARIA is recommended during the first 14 weeks of treatment with LEQEMBI. If a patient experiences symptoms suggestive of ARIA, clinical evaluation should be performed, including MRI if indicated. If ARIA is observed on MRI, careful clinical evaluation should be performed prior to continuing treatment.
- There is no experience in patients who continued dosing through symptomatic ARIA-E or through asymptomatic, but radiographically severe, ARIA-E. There is limited experience in patients who continued dosing through asymptomatic but radiographically mild to moderate ARIA-E. There are limited data in dosing patients who experienced recurrent ARIA-E.
Incidence of ARIA
- In Study 1 (Study 201), symptomatic ARIA occurred in 3% (5/161) of LEQEMBI-treated patients. Clinical symptoms associated with ARIA resolved in 80% of patients during the period of observation.
- Including asymptomatic cases, ARIA was observed in LEQEMBI: 12% (20/161); placebo: 5% (13/245). ARIA-E was observed in LEQEMBI: 10% (16/161); placebo: 1% (2/245). ARIA-H was observed in LEQEMBI: 6% (10/161); placebo: 5% (12/245). There was no increase in isolated ARIA-H for LEQEMBI compared to placebo.
- Intracerebral hemorrhage >1 cm in diameter was reported after one treatment in LEQEMBI: 1 patient; placebo: zero patients. Events of intracerebral hemorrhage, including fatal events, in patients taking LEQEMBI have also been reported in other studies.
Apolipoprotein E ε4 (ApoE ε4) Carrier Status and Risk of ARIA
- In Study 1, 6% (10/161) of patients in the LEQEMBI group were ApoE ε4 homozygotes, 24% (39/161) were heterozygotes, and 70% (112/161) were noncarriers.
- The incidence of ARIA was higher in ApoE ε4 homozygotes than in heterozygotes and noncarriers among patients treated with LEQEMBI. Of the 5 LEQEMBI-treated patients who had symptomatic ARIA, 4 were ApoE ε4 homozygotes, 2 of whom experienced severe symptoms. An increased incidence of symptomatic and overall ARIA in ApoE ε4 homozygotes compared to heterozygotes and noncarriers in LEQEMBI-treated patients has been reported in other studies.
- The recommendations on management of ARIA do not differ between ApoE ε4 carriers and noncarriers.
- Consider testing for ApoE ε4 status to inform the risk of developing ARIA when deciding to initiate treatment with LEQEMBI.
Radiographic Findings
- The majority of ARIA-E radiographic events occurred early in treatment (within the first 7 doses), although ARIA can occur at any time and patients can have more than 1 episode. The maximum radiographic severity of ARIA-E in patients treated with LEQEMBI was mild in 4% (7/161) of patients, moderate in 4% (7/161) of patients, and severe in 1% (2/161) of patients. Resolution on MRI occurred in 62% of ARIA-E patients by 12 weeks, 81% by 21 weeks, and 94% overall after detection. The maximum radiographic severity of ARIA-H microhemorrhage in patients treated with LEQEMBI was mild in 4% (7/161) of patients and severe in 1% (2/161) of patients; 1 of the 10 patients with ARIA-H had mild superficial siderosis.
Concomitant Antithrombotic Medication and Other Risk Factors for Intracerebral Hemorrhage
- Patients were excluded from enrollment in Study 1 for baseline use of anticoagulant medications. Antiplatelet medications such as aspirin and clopidogrel were allowed. If anticoagulant medication was used because of intercurrent medical events that required treatment for ≤4 weeks, treatment with LEQEMBI was to be temporarily suspended.
- Most exposures to antithrombotic medications were to aspirin; few patients were exposed to other antiplatelet drugs or anticoagulants, limiting any meaningful conclusions about the risk of ARIA or intracerebral hemorrhage in patients taking other antiplatelet drugs or anticoagulants. Because intracerebral hemorrhages >1 cm in diameter have been observed in patients taking LEQEMBI, additional caution should be exercised when considering the administration of antithrombotics or a thrombolytic agent (e.g., tissue plasminogen activator) to a patient already being treated with LEQEMBI.
- Patients were excluded from enrollment in Study 1 for the following risk factors for intracerebral hemorrhage: prior cerebral hemorrhage >1 cm in greatest diameter, more than 4 microhemorrhages, superficial siderosis, evidence of vasogenic edema, evidence of cerebral contusion, aneurysm, vascular malformation, infective lesions, multiple lacunar infarcts or stroke involving a major vascular territory, and severe small vessel or white matter disease. Caution should be exercised when considering the use of LEQEMBI in patients with these risk factors.
Infusion-Related Reactions
- Infusion-related reactions were observed in LEQEMBI: 20% (32/161); placebo: 3% (8/245), and the majority of cases in LEQEMBI-treated patients (88%, 28/32) occurred with the first infusion. All infusion-related reactions were mild (56%) or moderate (44%) in severity. Infusion-related reactions resulted in discontinuations in 2% (4/161) of patients treated with LEQEMBI. Symptoms of infusion-related reactions included fever and flu-like symptoms (chills, generalized aches, feeling shaky, and joint pain), nausea, vomiting, hypotension, hypertension, and oxygen desaturation.
- After the first infusion, 38% of LEQEMBI-treated patients had transient decreased lymphocyte counts to <0.9 x109/L compared to 2% on placebo, and 22% of LEQEMBI-treated patients had transient increased neutrophil counts to >7.9 x109/L compared to 1% on placebo.
- In the event of an infusion-related reaction, the infusion rate may be reduced, or the infusion may be discontinued, and appropriate therapy initiated as clinically indicated. Prophylactic treatment with antihistamines, acetaminophen, nonsteroidal anti-inflammatory drugs, or corticosteroids prior to future infusions may be considered.
ADVERSE REACTIONS
- In Study 1, 15% of LEQEMBI-treated patients, compared to 6% of placebo-treated patients, stopped study treatment because of an adverse reaction. The most common adverse reaction leading to discontinuation of LEQEMBI was infusion-related reactions that led to discontinuation in 2% (4/161) of patients treated with LEQEMBI compared to 1% (2/245) of patients on placebo.
- The most common adverse reactions reported in ≥5% of patients treated with LEQEMBI (N=161) and ≥2% higher than placebo (N=245) in Study 1 were infusion-related reactions (LEQEMBI: 20%; placebo: 3%), headache (LEQEMBI: 14%; placebo: 10%), ARIA-E (LEQEMBI: 10%; placebo: 1%), cough (LEQEMBI: 9%; placebo: 5%), and diarrhea (LEQEMBI: 8%; placebo: 5%).
Please see full Prescribing Information.
-
1
Alzheimer’s Association, 2022 Alzheimer's Disease Facts and Figures
https://www.alz.org/media/Documents/alzheimers-facts-and-figures.pdf -
2
Alzheimer’s Association, Changing the Trajectory of Alzheimer’s Disease
https://www.alz.org/media/documents/changing-the-trajectory-r.pdf -
3
Prados M, et al. Projecting the long‐term societal value of a disease‐modifying treatment for Alzheimer's disease in the United States. Alzheimers Dement. 2022; 18(1):142-151. doi: 10.1002/alz.12578. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9303743/
-
4
van Dyck. C, et al. Lecanemab in Early Alzheimer’s Disease. The New England Journal of Medicine. DOI: 10.1056/NEJMoa2212948.
https://www.nejm.org/doi/full/10.1056/NEJMoa2212948 -
5
Swanson CJ, et al. A randomized, double-blind, phase 2b proof-of-concept clinical trial in early Alzheimer’s disease with lecanemab, an anti-Aβ protofibril antibody
https://alzres.biomedcentral.com/articles/10.1186/s13195-021-00813-8 -
6
Kansal AR, Tafazzoli A, Ishak KJ, Krotneva S. Alzheimer's disease Archimedes condition-event simulator: Development and validation. Alzheimers Dement (NY). 2018;4:76-88. Published 2018 Feb 16. doi:10.1016/j.trci.2018.01.001
-
7
Tafazzoli A, and Kansal A. Disease simulation in drug development, External validation confirms benefit in decision making. The Evidence Forum. 2018.
https://www.evidera.com/wp-content/uploads/2018/10/07-Disease-Simulation-in-Drug-Development_Fall2018.pdf -
8
Tafazzoli A, Weng J, Sutton K, et al. Validating simulated cognition trajectories based on ADNI against 436 trajectories from the National Alzheimer's Coordinating Center (NACC) dataset. 11th edition of Clinical Trials on 437 Alzheimer's Disease (CTAD); Barcelona, Spain: 2018.
-
9
Tahami Monfared AA, et al. Long-term health outcomes of lecanemab in patients with early Alzheimer's disease using simulation modeling. Neurol Ther. 2022;11:863–880.
https://link.springer.com/article/10.1007/s40120-022-00350-y -
10
Tahami Monfared AA, et al. The Potential Economic Value of Lecanemab in Patients with Early Alzheimer’s Disease Using Simulation Modeling, Neurol Ther. 2022;11: 1285–1307.
https://link.springer.com/article/10.1007/s40120-022-00373-5 -
11
ICER Value Framework 2020-2023. 2022.
https://icer.org/wp-content/uploads/2020/11/ICER_2020_2023_VAF_02032022.pdf -
12
IQVIA. Longitudinal Access and Adjudication DATA (LAAD) 2019-2021
-
13
20% of the WAC price ($26,500) divided by 365 (days)
-
14
Internal Eisai calculations, December 2022
Safe Harbor Statement
Materials and information provided in this announcement may contain so-called “forward-looking statements.” These statements are based on current expectations, forecasts and assumptions that are subject to risks and uncertainties that could cause actual outcomes and results to differ materially from these statements.
Risks and uncertainties include general industry and market conditions, and general domestic and international economic conditions such as interest rate and currency exchange fluctuations. Risks and uncertainties particularly apply with respect to product-related forward-looking statements. Product risks and uncertainties include, but are not limited to, technological advances and patents attained by competitors; challenges inherent in new product development, including completion of clinical trials; claims and concerns about product safety and efficacy; regulatory agency examination periods and obtaining regulatory approvals; domestic and foreign healthcare reforms; trends toward managed care and healthcare cost containment; and governmental laws and regulations affecting domestic and foreign operations.
The Company cannot guarantee the actual outcomes and results for any forward-looking statements.
The Company disclaims any intention or obligation to update or revise any forward-looking statements whether as a result of new information, future events or otherwise.