- For Print
- September 18, 2020
Eisai Co., Ltd. (Headquarters: Tokyo, CEO: Haruo Naito, “Eisai”) announced that the latest results from the cohort targeting patients with HER2-negative breast cancer in the phase I clinical trial for the new liposomal formulation (E7389-LF) of the in-house discovered anti-cancer treatment Halaven® (generic name: eribulin mesylate, “eribulin”) were presented (abstract number: 346P) at the European Society for Medical Oncology (ESMO) Virtual Congress 2020.
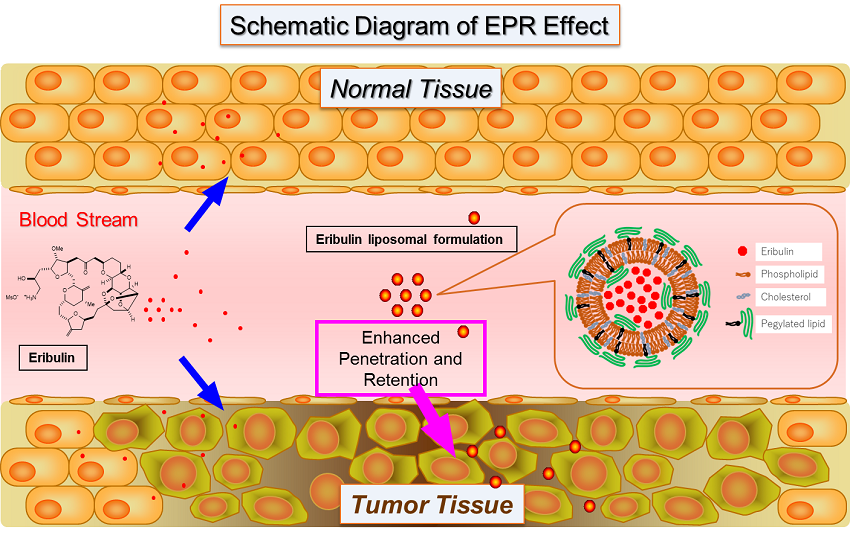
E7389-LF is a new formulation using liposomes made of a lipid bilayer to encapsulate the halichondrin-class microtubule dynamics inhibitor eribulin. In tumor tissue, gaps exist among vascular endothelial cells due to incomplete vasculature, which is thought to allow for penetration by macromolecules. This condition, in addition to incomplete lymphatic function, is predicted to enable high-molecular-weight drugs including liposomal formulations to be respectively delivered and retained in greater amounts in tumors as compared to in normal tissue, through Enhanced Permeability and Retention (EPR) effects. Thus E7389-LF is expected to improve the concentration of eribulin in tumor tissues.
This presentation reported efficacy and safety results of E7389-LF in a cohort enrolling 28 patients with recurrent (HER2-negative) breast cancer (hormone receptor positive: 21, triple negative: 7) who had previously undergone treatment with anthracycline or taxane class treatments and had no prior treatment with eribulin (data cutoff: January 24, 2020, progression free survival and overall survival cutoff: April 17, 2020), as part of the open-label, phase I clinical trial (Study 114) on patients with select solid tumors who had previously undergone treatment. Patients were treated with E7389-LF 2.0 mg/m2 body surface area (as free eribulin) intravenously once every three weeks, and demonstrated an overall response rate (ORR) of 35.7% (95% confidence interval (CI): 18.6-55.9) in the HER2-negative breast cancer cohort as a whole. Within the cohort, hormone receptor positive patients demonstrated an ORR of 42.9% (95% CI: 21.8-66.6) and triple negative patients demonstrated an ORR of 14.3% (95% CI: 0.4-57.9). The disease control rate combining stable disease rate, partial response rate, and complete response rate was 89.3% (95% CI: 71.8-97.7). Additionally, progression-free survival (PFS) was a median of 5.7 months (95% CI: 3.9-8.3), and the median overall survival (OS) was not reached (95% CI: 10.3 – not reached). Adverse events of grade 3 or above (top 5) were neutropenia (67.9%), leukopenia (42.9%), thrombocytopenia (32.1%), febrile neutropenia (25.0%), and increased alanine aminotransferase (21.4%), which were consistent with the safety profile of eribulin to date. Additionally, preventive treatment with the G-CSF (granulocyte colony stimulating factor) pegfilgrastim demonstrated a decrease in the ratio of febrile neutropenia occurrence (with treatment: 10.0%, without treatment: 33.3%).
Eisai positions oncology as a key franchise area and aims to create innovative drugs that act towards curing cancer. Eisai will continue to create innovation in the development of new drugs based on cutting-edge cancer research, and aims to make continuous efforts to meet the diversified needs of and increase the benefits provided to patients with cancer, their families, and healthcare professionals.
Media Inquiries:
Public Relations Department,
Eisai Co., Ltd.
+81-(0)3-3817-5120
[Notes to editors]
- 1. About E7389-LF
E7389-LF is a new formulation designed for more efficient delivery of the halichondrin-class microtubule dynamics inhibitor “Halaven” (Eribulin mesylate) into cancer cells by liposome envelopment. A phase I clinical study is currently being conducted on select solid tumors in Japan. Additionally, a phase Ib/II clinical trial on the combination therapy of E7389-LF and nivolumab targeting select solid tumors is currently being conducted in Japan in collaboration with Ono Pharmaceutical Co., Ltd.
- 2. About Halaven (generic name: eribulin mesylate)
Halaven is in the halichondrin class of microtubule dynamics inhibitors with a novel mechanism of action. Structurally, Halaven is a simplified and synthetically produced version of halichondrin B, a natural product isolated from the marine sponge Halichondria okadai. Halaven is believed to work by inhibiting the growth phase of microtubule dynamics which prevents cell division. In addition, non-clinical studies showed Halaven’s unique actions in the tumor microenvironment such as an increase in vascular perfusion and permeability in tumor cores1, promotion of the epithelial state, decrease in capacity of breast cancer cells to migrate2, etc.
Halaven was first approved as a treatment in the United States in November 2010 for patients with metastatic breast cancer. Halaven is currently approved for use in the treatment of breast cancer in over 75 countries worldwide, including Japan, China and countries in Europe, the Americas and Asia. Furthermore, Halaven was first approved as a treatment for soft tissue sarcoma in the United States in January 2016, and is approved in over 65 countries including Japan and in Europe and Asia. Furthermore, Halaven has been designated as an orphan drug for soft tissue sarcoma in the United States and Japan.
Specifically, Halaven is approved for the following indications.
In the United States for the treatment of patients with:
- Metastatic breast cancer who have previously received at least two chemotherapeutic regimens for the treatment of metastatic disease. Prior therapy should have included an anthracycline and a taxane in either the adjuvant or metastatic setting.
- Unresectable or metastatic liposarcoma who have received a prior anthracycline-containing regimen.
In Japan for the treatment of patients with:
- Inoperable or recurrent breast cancer, soft tissue sarcoma
In Europe for the treatment of adult patients with:
- Locally advanced or metastatic breast cancer who have received a prior anthracycline-containing regimen for advanced disease. Prior therapy should have included an anthracycline and a taxane in either the adjuvant or metastatic setting, unless patients were not suitable for these treatments.
- Unresectable liposarcomas who have received prior anthracycline containing therapy (unless unsuitable) for advanced or metastatic disease.
Two open-label, randomized, phase III trials (EMBRACE and Study 301) were conducted for Halaven in patients with locally advanced or metastatic breast cancer previously treated with an anthracycline and a taxane. Based on the pooled analysis of the combined results3, overall survival (OS) was extended significantly in the Halaven group compared to the control group (Hazard Ratio 0.85 [95% Confidence Interval (CI) = 0.77-0.95] p = 0.003, Halaven median OS: 15.2 months vs. control median OS: 12.8 months). Progression free survival (PFS) was also extended in the Halaven group compared to the control group (Hazard Ratio 0.90 [95% CI = 0.81-0.997] p = 0.046, Halaven median PFS: 4.0 months vs. control median PFS: 3.4 months).
The overall response rate (ORR) in the HER2-negative breast cancer group was 13.5%. Within the group, an ORR of hormone receptor positive patients was 14.3% and that of triple negative patients was 12.0%. Median PFS in the HER2-negative breast cancer group was 4.0 months.
Regarding the safety profile from the combined analysis, there were no major differences among the respective clinical studies. Patients in these phase III studies received Halaven (1.4 mg/m2 administered intravenously on Day 1 and Day 8) every 21 days.
-
1
Funahashi Y et al., Eribulin mesylate reduces tumor microenvironment abnormality by vascular remodeling in preclinical human breast cancer models. Cancer Sci., 2014; 105, 1334-1342
-
2
Yoshida T et al., Eribulin mesilate suppresses experimental metastasis of breast cancer cells by reversing phenotype from epithelial-mesenchymal transition (EMT) to mesenchymal-epithelial transition (MET) states. Br J Cancer, 2014; 110, 1497-1505
-
3
Twelves C et al., Efficacy of eribulin in women with metastatic breast cancer: a pooled analysis of two phase 3 studies. Breast Cancer Res Treat, 2014; 148, 553-561
Our news releases are intended to disclose corporate information and are not intended for the promotion or advertising of prescription pharmaceuticals or investigational products, nor are they intended to provide medical advice.
Please note that the information contained in news releases is current as of the date of publication.
