- For Print
- July 11, 2018
Sato Pharmaceutical Co., Ltd.
Eisai Co., Ltd.
Sato Pharmaceutical Co., Ltd. (Headquarters: Tokyo, President and CEO: Seiichi Sato, “Sato Pharmaceuical”) and Eisai Co., Ltd. (Headquarters: Tokyo, CEO: Haruo Naito, “Eisai”) will launch the oral antifungal agent NAILIN® Capsules 100mg in Japan on July 27, 2018.
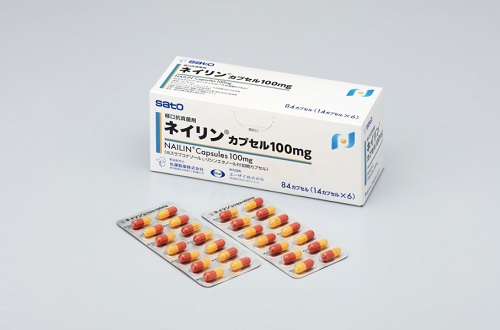
Sato Pharmaceutical obtained marketing and manufacturing approval for NAILIN® Capsules 100mg as an oral treatment for onychomycosis. This product is the first new treatment for the disease in approximately 20 years. NAILIN® Capsules 100mg contains the active ingredient fosravuconazole L-lysine ethanolate which was discovered by Eisai and has improved bioavailability compared to ravuconazole.
Caused by the invasion of the nails by the Trichophyton fungus via the skin of the hands and feet, onychomycosis is a fungal infection that induces symptoms such as the clouding and thickening of nails as well as hyperkeratosis in the area surrounding the nail. Onychomycosis affects 1 in every 10 Japanese people with an estimated approximately 11 million sufferers in Japan, and incidence rises with age. Although it is an easy disease to leave untreated as it is not associated with pain or itchiness, if the disease progresses, various symptoms may occur such as pain from misshapen nails, difficulty in putting on shoes and pain upon walking, and many patients are concerned about reduction in QOL (quality of life). Furthermore, it is possible for Trichophyton to infect other people by sharing slippers, bathmats and other mediums.
In a Phase III clinical study conducted by Sato Pharmaceutical of NAILIN® Capsules 100mg administered orally once daily for 12 weeks in patients with onychomycosis conducted in Japan, superior efficacy was confirmed for NAILIN® Capsules 100mg compared to placebo.
Going forward, Sato Pharmaceutical will conduct marketing of NAILIN® Capsules 100mg, while Sato Pharmaceutical and Eisai are jointly providing information on its proper use. By expanding the options for treating onychomycosis through NAILIN® Capsules 100mg, the two companies are striving to further contribute to the treatment of onychomycosis patients.
(Outline of NAILIN® Capsules 100 mg)
[Date Listed in the Drug Pricing Standard System]
May 22, 2018
[Price]
804.60 yen (100 mg per capsule)
[Date of Launch]
July 27, 2018
[Date of Marketing and Manufacturing Authorization]
January 19, 2018
[Product Name]
NAILIN® Capsules 100 mg
[Active Ingredient]
Fosravuconazole L-lysine ethanolate
[Indication]
<Indicated Type of Bacteria> Dermatophytosis (Trichophyton genus)
<Indicated Disease> Onychomycosis
[Dosage and Administration]
The usual adult dosage is 1 capsule (100 mg as ravuconazole) administered orally once daily for a period of 12 weeks
[Packaging]
84 capsules (PTP 14 capsules X 6)
Media Inquiries
-
Sato Pharmaceutical Co., Ltd.
Public Relations Department
-
Eisai Co., Ltd.
Public Relations Department
[Notes to editors]
1. About Sato Pharmaceutical Co., Ltd.
Sato Pharmaceutical Co., Ltd., operating under its corporate philosophy of “Healthcare Innovation”, is a pharmaceutical company that provides effective, safe, and highquality products for practicing selfcare, while always keeping the health of its customers in mind. In addition to its main consumer healthcare business, Sato Pharmaceutical also develops and provides highly original products primarily in the field of dermatology.
Regarding ethical pharmaceuticals, through the marketing of NAILIN® Capsules 100 mg in addition to the onychomycosis treatment LUCONAC® Solution 5% launched on April 25, 2016, Sato Pharmaceutical is able to provide both externally and internally administered medicines as options for the treatment of onychomycosis. Going forward, Sato Pharmaceutical is expanding promotion aimed at establishing a brand with these products as two pillars of onychomycosis treatment, and is striving to continue growing as a leading company in the treatment of onychomycosis that is trusted by healthcare professionals.
2. About Eisai Co., Ltd.
Eisai Co., Ltd. is a leading global research and development-based pharmaceutical company headquartered in Japan. We define our corporate mission as “giving first thought to patients and their families and to increasing the benefits health care provides,” which we call our human health care (hhc) philosophy. With approximately 10,000 employees working across our global network of R&D facilities, manufacturing sites and marketing subsidiaries, we strive to realize our hhc philosophy by delivering innovative products to address unmet medical needs, with a particular focus in our strategic areas of Neurology and Oncology.
Our news releases are intended to disclose corporate information and are not intended for the promotion or advertising of prescription pharmaceuticals or investigational products, nor are they intended to provide medical advice.
Please note that the information contained in news releases is current as of the date of publication.
