Advances in biology, information science, and digital technologies are enabling us to identify physical changes in the body even before a disease begins. As a result, healthcare is shifting from treating diseases after they appear to preventing them by detecting and managing these early changes in advance. In this approach, it is essential to view disease not as a single event but as an ongoing process (Disease Continuum). By understanding this process and its underlying causes, we can more precisely grasp the wide variety of disease mechanisms and develop more effective, tailored treatments.
DHBL (Deep Human Biology Learning)-based drug development is our proprietary R&D approach, which begins with a deep understanding of biological changes in the human body—such as disease-related genetic information, cellular functions, and pathophysiology (Human Biology).
A Message from Vice President, Chief Scientific Officer, Katsutoshi Ido

Eisai has a long-standing track record of drug development in the areas of neurology and oncology. Each year, we conduct more than 500 human health care (hhc) activities, and the insights gained through direct engagement with patients continue to inspire our researchers. This strong commitment to understanding and addressing patient needs has led to the creation of innovative treatments such as ARICEPT, FYCOMPA, and DAYVIGO in neurology, and HARAVEN, LENVIMA, and TASFYGO in oncology. In addition, we have successfully delivered LEQEMBI, a treatment for early Alzheimer’s disease (AD), by bringing together our core strengths. These strengths include a focus on non-clinical disease models that underpin our drug discovery concepts, craftsmanship across diverse therapeutic modalities, advanced biomarker research enabled by molecular profiling technologies, and clinical development focused on selecting appropriate patient populations. All of this has been driven by our commitment to the hhc philosophy and strong leadership.
LEQEMBI selectively binds to amyloid-beta (Aβ) protofibrils, which are considered a toxic species of Aβ and one of the key components of AD pathology. It reduces both soluble and insoluble Aβ aggregates in the brain, thereby slowing the progression of mild cognitive impairment (MCI) and mild AD.
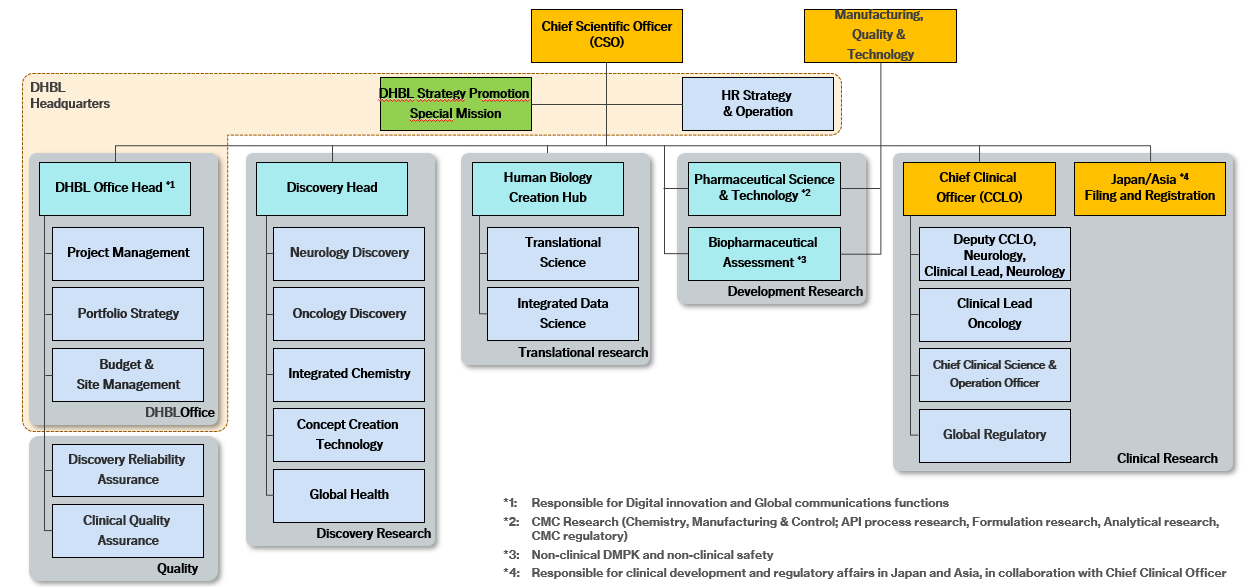
Organizational Structure of DHBL
The DHBL division, established in October 2022, embodies Eisai’s proprietary R&D framework based on human biology data as its starting point. To strengthen our mid- to long-term R&D capabilities, we refined this framework in FY2025 to promote more rigorous, science-driven discussions at all times and to enhance the strategic and efficient use of our core platform technologies, thereby improving research productivity. To continuously create value for patients, we accelerate our drug development activities by emphasizing swift and well-informed decision-making, optimal allocation of resources, and transparent information sharing (Figure 1).
Under the leadership of the Chief Scientific Officer (CSO) within the DHBL framework, the Human Biology Creation Hub (Figure 2) generates and validates novel drug discovery hypotheses, strengthens the integration of non-clinical and clinical research, accelerates clinical trials, and improves the likelihood of success through biomarker-driven approaches. It also advances the development of diagnostic technologies in partnership with the medical department.
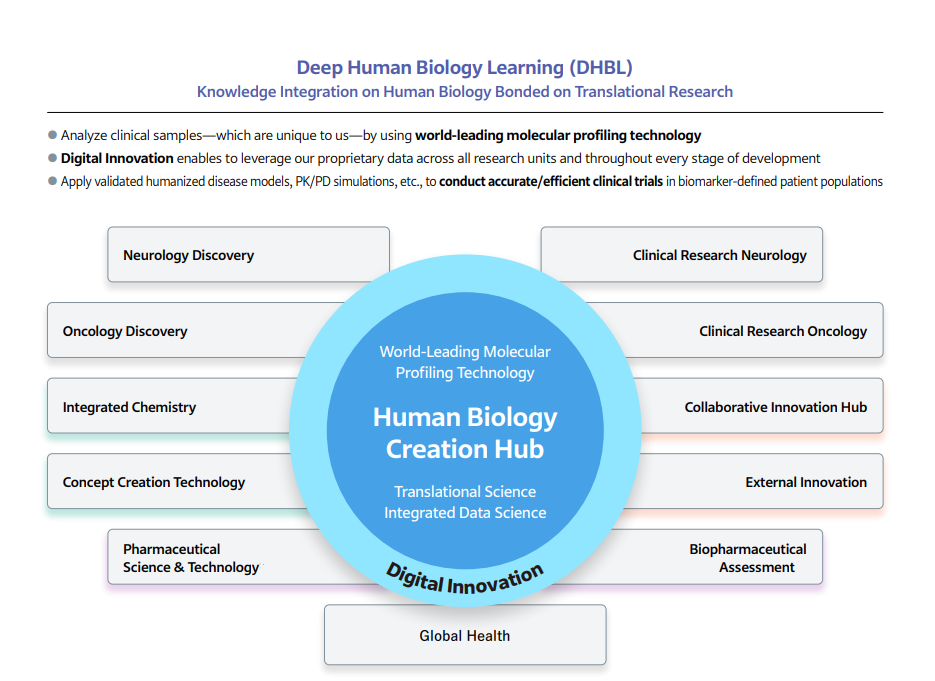
Discovery research focuses on two core therapeutic areas—neurology and oncology—as well as global health. The Neurology, Oncology, and Global Health Discovery function, composed of experts in non-clinical pharmacology, works closely with Integrated Chemistry, which leverages cutting-edge technologies such as artificial intelligence (AI), machine learning (ML), and structural biology. It also collaborates with the Concept Creation Technology function, which pioneers new drug modalities and develops advanced experimental validation capabilities. Through this integrated approach, we generate and rigorously assess high-confidence drug discovery hypotheses. By rapidly repeating this validation cycle, we accelerate the advancement of discovery projects into early clinical development. In addition, we actively engage in collaborative research with academia and biotechnology ventures (external innovation) to access cutting-edge technologies and advance partnerships with external collaborators.
The DHBL headquarters (DHBL HQ) is responsible for developing and driving portfolio strategies and optimizing R&D resource allocation while managing overall R&D functions to ensure that each research function contributes to achieving regulatory approvals as quickly and efficiently as possible. Additionally, DHBL HQ actively promotes digital innovation by leveraging R&D IT, AI, ML, and human biology data generated through our clinical research to pursue unique drug discovery approaches. To enhance researcher engagement and support their growth, we emphasize active communication. We also foster both a risk-taking mindset that embraces innovative and progressive challenges and a culture that values fast, well-grounded decision-making.
Driven by strong leadership and a shared sense of mission grounded in the hhc philosophy, we are united as a single “Monolith,” with all members working as one team to pursue our unwavering mission: to deliver innovative new medicines to patients and people in their daily lives as quickly, efficiently, and sustainably as possible, and to help alleviate health-related concerns. Looking ahead, we will continue to leverage the strengths of the DHBL framework and the capabilities we have built over time to accelerate drug development for patients and the public. We remain fully committed to these efforts and sincerely appreciate your continued support.

