- For Print
- May 28, 2012
Eisai Co., Ltd. (Headquarters: Tokyo, President & CEO: Haruo Naito, “Eisai”) announced today that its U.K. subsidiary Eisai Europe Ltd. has received a positive opinion from the European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) for the use of FycompaTM (perampanel) for the adjunctive treatment of partial-onset seizures, with or without secondarily generalized seizures, in patients with epilepsy aged 12 years and older.
Perampanel, a novel chemical entity discovered and in development by Eisai, is a highly selective, non-competitive AMPA-type glutamate receptor antagonist. If approved, perampanel will be the first product in this new class of anti-epileptic drugs to gain regulatory approval.
The CHMP based its decision on clinical data from 3 pivotal Phase III, global, randomized, double-blind, placebo-controlled, dose-escalation studies in 1,480 epilepsy patients. Each of the studies showed consistency with the preferable results in seizure control as adjunctive therapy across all partial onset seizure types, including secondary generalization. The most commonly reported adverse events were dizziness, headache, somnolence, irritability, fatigue, falls, and ataxia. According to the CHMP positive opinion, perampanel is expected to contribute to patients with epilepsy aged 12 years and older, that is adults and adolescents. Additionally, perampanel delivers the benefit of once-daily dosing, thereby facilitating adherence to treatment. Based on CHMP’s recommendation, EU approval of the new therapy is anticipated within three months.
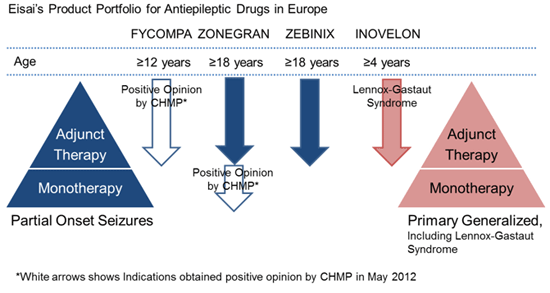
Eisai defines epilepsy as a therapeutic area of focus, and seeks to make further contributions to address the diversified needs of, and increase the benefits provided to, epilepsy patients and their families by providing them with multiple treatment options as part of its abundant epilepsy franchise product portfolio.
[ Please refer to the following notes for further information on epilepsy, AMPA receptor antagonist FycompaTM (perampanel), perampanel Phase III studies, and Eisai’s Commitment to Epilepsy ]
Media Inquiries:
Public Relations Department,
Eisai Co., Ltd.
+81-(0)3-3817-5120
< Notes to editors >
1. About Epilepsy
Epilepsy is a medical condition that produces seizures affecting a variety of mental and physical functions. A patient is considered to have epilepsy after two or more unprovoked seizures. A seizure occurs when a brief, strong surge of electrical activity affects part or all of the brain. An individual can have various symptoms, from convulsions and loss of consciousness, to some that are not always recognized as seizures, such as blank staring, lip smacking, or jerking movements of arms and legs.
Epilepsy can develop at any age and 0.5% to 2% of people will develop epilepsy during their lifetime. Epilepsy reportedly affects nearly 1 million people in Japan, 2.4 million people in Europe (G5), 3 million people in the United States, and more than 50 million people worldwide. Despite a wide range of therapeutic options, the treatment of partial-onset seizures (the most common type of epilepsy) remains a challenge. Currently, up to a third of people with epilepsy in Europe do not achieve seizure freedom despite therapy with anti-epileptic drugs.
2. About AMPA Receptor Antagonist FycompaTM (Perampanel)
FycompaTM (Perampanel), a novel chemical entity discovered and in development by Eisai, is a highly selective, non-competitive AMPA -type glutamate receptor antagonist. Perampanel is the first anti-epileptic treatment to reduce neuronal hyperexcitation associated with seizures by targeting glutamate activity at post-synaptic AMPA receptors. Perampanel has demonstrated broad-spectrum anti-seizure effects in Phase II and III studies. The agent has received a positive opinion from the CHMP for the adjunctive treatment of partial-onset seizures in Europe, is currently under regulatory review for NDA in the United States, and is also being evaluated in phase III studies in Japan. Furthermore, Eisai is conducting a global phase III studies for generalized epilepsy and plans to conduct further studies for usage as monotherapy in the treatment of partial-onset seizures, Lennox-Gastaut syndrome and other forms of epilepsy as it seeks to expand the range of indications for which the drug is approved.
3. About Perampanel Phase III studies
The clinical development plan for perampanel consisted of three global Phase III studies: Studies 306, 305 and 304 in which a total of 1,480 epilepsy patients aged 12 years and older participated. The key goal of Study 306 was to identify the minimal effective dose and included four treatment arms (placebo, 2mg, 4mg, and 8mg). Studies 304 and 305 included three arms (placebo, 8mg, and 12mg) and were to evaluate a more extended dose range.
The studies were similar in design: global, randomised, double-blind, placebo-controlled, dose-escalation, parallel-group studies. The primary and secondary endpoints were the same in all the studies: percentage change in seizure frequency, 50% responder rate, percentage reduction of complex partial plus secondarily generalized seizures, and evaluation for dose response. The primary endpoint for the EMA is 50% responder rate and the FDA is median percent change in seizure frequency. Specifically the results showed:
Study 306
- The 50% responder rates compared to placebo for the ITT (intention-to-treat) population were: 20.6% (p=0.4863), 28.5% (p=0.0132), and 34.9% (p=0.0003) in the 2, 4, and 8 mg perampanel/day groups, respectively, versus 17.9% with placebo.
- The median percent change in seizure frequency for the ITT population shown: 2 mg = -13.6% (p=0.4197), 4 mg = -23.3% (p=0.0026), 8 mg = -30.8% (p<0.0001) and placebo = -10.7%
- The most frequent treatment-emergent adverse events were dizziness, headache and somnolence.
Study 305
- The 50% responder rates compared to placebo for the ITT (intention-to-treat) population were:, 33.3% (p=0.0018), and 33.9% (p=0.0006) in the 8 and 12 mg perampanel/day groups, respectively, versus 14.7% with placebo.
- The median percent change in seizure frequency for the ITT population shown: 8mg = -30.5% (p=0.0008), 12mg = -17.6% (p=0.0105) and placebo = -9.7%
- The most reported adverse events were dizziness, fatigue, headache and somnolence
Study 304
- The 50% responder rates compared to placebo for the ITT (intention-to-treat) population were: 37.6% (p=0.0760), and 36.1% (p=0.0914) in the 8 and 12 mg perampanel/day groups, respectively, versus 26.4% with placebo.
- The median percent change in seizure frequency for the ITT population shown: 8mg = -26.3% (p=0.0261), 12mg = -34.5% (p=0.0158) and placebo = -21.0%
- The most common side effects were dizziness, somnolence, irritability, headache, falls, and ataxia.
4. Eisai’s Commitment to Epilepsy
Eisai defines epilepsy as a therapeutic area of focus, not only developing FycompaTM (Perampanel) globally, but currently marketing Zonegran® (under license from the originator Dainippon Sumitomo Pharma Co., Ltd.; sodium/calcium channel blocking antiepileptic agent; marketed in Europe, the United States and Asia) and Zebinix® (under license from the originator BIAL-Portela & Ca S.A.; voltage-dependent sodium channel-blocking antiepileptic agent; marketed in Europe) as adjunctive therapies in adults with partial onset seizures, and Inovelon®/BANZEL® (under license from the originator Novartis AG; sodium channel-blocking novel triazole derived antiepileptic agent; marketed in Europe, Asia (Inovelon®), and the North America (BANZEL®) for the adjunctive treatment of seizures associated with Lennox-Gastaut syndrome, a severe form of early childhood-onset epilepsy. In May 2012, Eisai also received a positive CHMP opinion for Zonegran® as monotherapy for the treatment of partial seizures with newly diagnosed epilepsy.