October, 2013
Takahiro Ogawa
Immunology & Inflammation Section, Integrated Marketing Department
Juvenile idiopathic arthritis (childhood arthritis or “JIA”), which is designated as a chronic pediatric disease in Japan, places a heavy burden on patients and their families and creates uncertainty about the future. Eisai worked to overcome hurdles associated with pediatric clinical trials and in July 2011 its antibody drug Humira® was approved by the Japanese Ministry of Health, Labour and Welfare as a treatment for this disease. Since then, we have been working to deliver “hope for the future” to patients living with JIA. Being able to see firsthand the contribution that Humira is making in the lives of these children, including one young person who was able to gain a new lease on life through successful treatment and go on to medical college to pursue dreams of becoming a doctor, is one of the aspects of pediatric medicine that make this field so rewarding.
On the other hand, JIA is a relatively rare disease and not yet widely known. The reality is that even when the initial symptoms are present, pediatric patients may not always be able to fully express what is happening, family members may lack understanding about the disease, and doctors may fail to accurately diagnose JIA in its early stages. To remedy this situation, Eisai has been working to raise JIA awareness through various initiatives such as organizing lecture meetings for orthopedists, who are often the first to encounter the initial symptoms of the disease, and creating “web seminars” for distribution to medical practitioners.
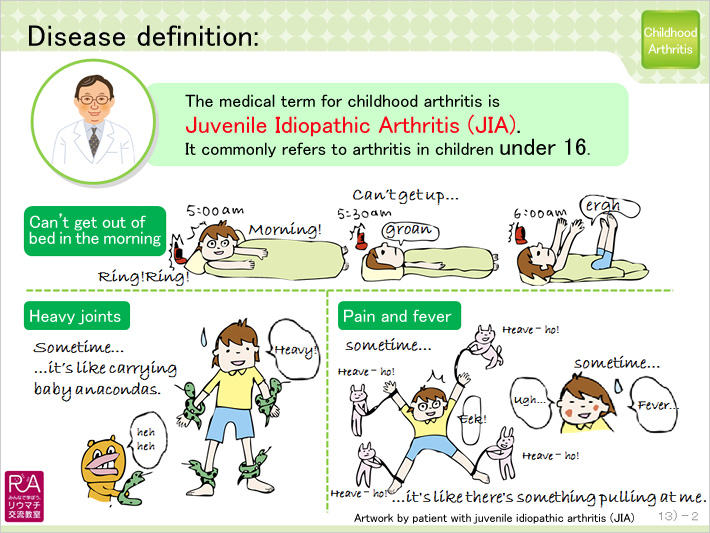
Eisai has also been creating JIA slide kits that include symptom illustrations of patients' experiences living with the disease as depicted by the children themselves. The kits aim to explain JIA symptoms and management in a way that can be readily understood by children and family members alike. Furthermore, the slide kits also serve to share the experiences of patients with JIA in a way that can help other children living with the disease.
Prof. Syuji Takei is the President of the Pediatric Rheumatology Association of Japan (PRAJ), which has been collaborating with Eisai to create the slide kits. He comments: “Working to better understand the emotions and experiences of children with JIA and their families is just the sort of initiative that I've come to expect from Eisai. I look forward to seeing more activities by Eisai as a part of their hhc philosophy.”
Eisai will continue to strive to deliver “hope for the future” to as many pediatric patients and their families as possible, not only through treatments but initiatives to improve access to medicines as well, in order to provide further benefits to children living with JIA and their families.
- *PRAJ post held by Prof. Takei current at time of interview and at project's implementation.